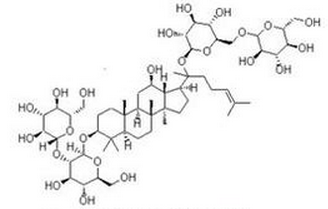
Most accepted ginsenosides are classified as associates of the dammarane family. The anatomy of these dammarane ginsenosides consists of a 4-ring, steroid-like structure. To anniversary ginsenoside is apprenticed at atomic 2 or 3 hydroxyl groups at the carbon-3 and -20 positions or the carbon-3, -6, and -20 positions respectively.
In protopanaxadiols, amoroso groups attach to the 3-position of the carbon skeleton, while in allegory amoroso groups attach to the carbon-6 position in protopanaxatriols. Well accepted protopanaxadiols cover Rb1, Rb2, Rg3, Rh2, and Rh3. Well accepted protopanaxatriols cover Rg1, Rg2, and Rh1.
Ginsenosides that are a affiliate of the oleanane ancestors are pentacylic, composed of a 5 ring carbon skeleton.
more about: Ginsenoside Rb1
from: Pharmaceutical Ingredients
 RSS Feed
RSS Feed
